–Chris Robbins, Jamaica
We all can remember the model of atomic structure taught us in high school chemistry and physics classes, something similar to a planet with many moons. The protons and neutrons were clumped together, forming the planet and the electrons were arranged in orderly orbits (valence rings). This is a useful model for teaching atomic structure and chemical interactions, and it is an accurate model of the average distribution of electrons over a period of time. But, instantaneously, reality is much messier.
Electrons are in constant motion within and among atoms of a molecule. At any given instant, their cumulative charge may be arranged asymmetrically, making molecules dipoles with north and south poles similar to that of bar magnets. These dipoles tend to align themselves with the magnetic fields of passing electromagnetic (EM) waves. In the right frequency band in which EM waves oscillate sufficiently rapidly so that there are many cycles during the period, a dipole orientation is stable, the dipoles oscillate sympathetically with the EM waves’ magnetic field. This creates friction and the resulting heat. At frequencies below this range, dipoles don’t remain in any given orientation long enough to vibrate. Above it, the inertia of molecule dipoles limit their sympathetic motion.
Microwave ovens exploit this phenomenon to cook our food, primarily oscillating the water molecules in food to heat it. Molecules have mass as well as momentary dipole magnetic fields. If the electromotive force of a passing EM wave is too small to accelerate the molecule’s mass, nothing happens: no vibration, friction, or heat. Limits on power and the oven’s screening limit microwave oven external radiation to harmless levels unable to move molecules. They can’t hurt us.
Fortunately, EM waves with field strengths at or below those outside our microwave ovens are still strong enough to move the electrons in antennas thereby creating detectable voltages that can be amplified, digitized, and processed into the information the Internet brings us. 2.45 GHz is the standard frequency for microwave ovens, a frequency that is right in the middle of the middle band of 5G frequencies. This may be why there is concern for potential harmful effects from 5G EM radiation. While the frequency is potentially of concern, the power levels, like those outside your microwave oven, are too low to harm us.
The atoms of molecules in our skin, mostly water, are held together with forces that are elastic, i.e., they can vibrate. At higher frequencies in the infrared, visible light, and ultraviolet range, these bonds can vibrate in resonance with the frequency of passing EM waves, again causing friction and heat. In the infrared (IR) band, this warms us. In the visible light band, some of the energy in the EM waves is absorbed and some is reflected, making us visible. And, in the ultra violet range, some is absorbed as vitamin C. In this band, the radiation is essential for life, but in excess, it can be harmful. We are so familiar with these effects that there really are no unfounded concerns.
At even higher frequencies in the X-ray and Gama ray bands, EM waves can be ionizing. That is, they can knock electrons of our cells’ DNA molecules loose thereby damaging or killing the cell. As with other frequency bands, whether EM waves are useful or harmful depends on their power level. At low power levels, X-rays are used for medical imaging and Gamma rays to treat cancer. At higher levels or amounts accumulated over time, they can harm or kill us.
How can we be sure electromagnectic waves are too weak to hurt us?
There are two primary mechanisms that decrease the strengths of EM waves as they propagate from their source: spreading loss and attenuation. Spreading loss can be illustrated by considering what happens when a stone is thrown into a pond. The splash creates a water wave that spreads in a circle from the splash. As the wave spreads, the energy spreads over the expanding circumference and diminishes proportionally. The wave amplitude at any point of the expanding circle is inversely proportional to the amplitude of the original splash by the circumference of the circle (1/2πR where π is the ratio of a circle’s circumference to its diameter and R is the circle’s radius). This is an example of circular spreading loss.
EM waves diminish as they spread or propagate, too, except that they spread spherically in all directions instead of circularly. Therefore, at any distance, the energy at the EM’s source is spread over the area of a sphere with that distance as its radius and is inversely proportional to the source strength by the ratio of the sphere surface area (1/(4/3)πR2). By convention, spreading losses of EM waves are measured by the ratio of power at a given distance to that at a sphere of 1 meter radius that reduces to 1/R2. So, at 10 meters, EM strength is diminished 40 db, at 100 meters 80 db, and at a km, 120 db , i.e., 20 log (1/R2).
Spreading loss can be experienced using a flashlight. Hold it an inch or so from your eye and shine it directly into your pupil. The light will be so intense that it will blind you to everything else. Now move it to arm’s length and shine it directly into the same pupil. It may be bright, but not so bright that you won’t be able to see everything else in your field of vision. The difference in intensity is spreading loss, about 1/1000 of the near intensity or a 60 db decrease. Light is EM waves at frequencies 10,000 times that of 5G. But, they obey the same spreading loss law.
The second loss mechanism, attenuation, depending on frequency, is much less severe in air. At 5G frequencies, it is less than 1 db per km of distance. In water, it is far more severe which is why rain can interfere with EM propagation and if heavy enough, block it altogether. These losses put 5G EM waves in the ‘goldielocks” region of being too weak to effect us, but more than strong enough for the sophisticated antennas and electronics in our devices to detect and process.
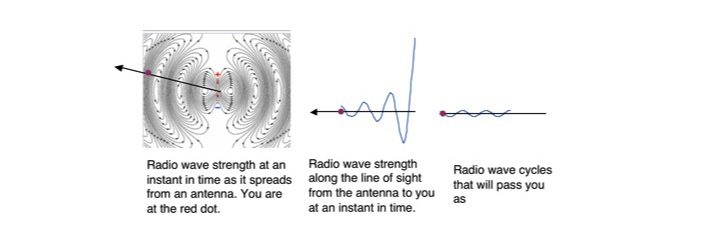
Figure 1
Figure 1. The strength of a radio wave in a horizontal plane at an instant in time is shown on the left. Gray is a wave peak and white a valley. A line of sight to an observer is shown. The distance between intense gray areas is the wavelength. The middle figure shows the decrease in radio wave strength at an instant in time along the line of sight to the observer as it has spread out from the antenna. The figure to the right is what is what the observer experiences as the wave passes. The number of peaks that pass in one second is the frequency.
A final thought, at the foot of cell towers where slant range to the broadcast antenna may not be that great, how can we be sure that EM wave strength is diminished sufficiently to be harmless? Cell towers broadcast EM waves in beams. EM wave strength is concentrated in a beam’s direction and decreased it in all others. Spreading loss at any point of the beam’s wave front is still spherical relative to the source, but it is not uniform over the arc of the wave front. Cellular beams are oriented horizontally so power at the foot of the tower is less than the power further from the tower where the beams intercept the ground. A visible example of this is can be seen with a flashlight beam. To see this phenomenon, hold a flashlight exactly perpendicular to your line of sight. You may see some reflection from dust particles in the air, but you won’t see the beam. Now rotate the flashlight until it shines in your face. The increasing brightness as it rotates is a measure of its beam’s intensity pattern. EM wave beams at 5G frequencies behave the same way and at angles that are nearly perpendicular to their axis they are far too weak to harm us.
Sources
1. Wikipedia 5G
2. Propagation; https://m.eet.com/media/1115910/mcclaning_3_pt1.pdf
3. Basic Properties of Radiation Biology; Bookshelf; https://www.ncbi.nlm.nih.gov/books/NBK232435/
4. How does light, infrared and UV radiation interact with skin and eyes?; GreenFacts for Life and Health; https://copublications.greenfacts.org/en/energy-saving-lamps/l-3/2-light-interaction-skin-eye.htm